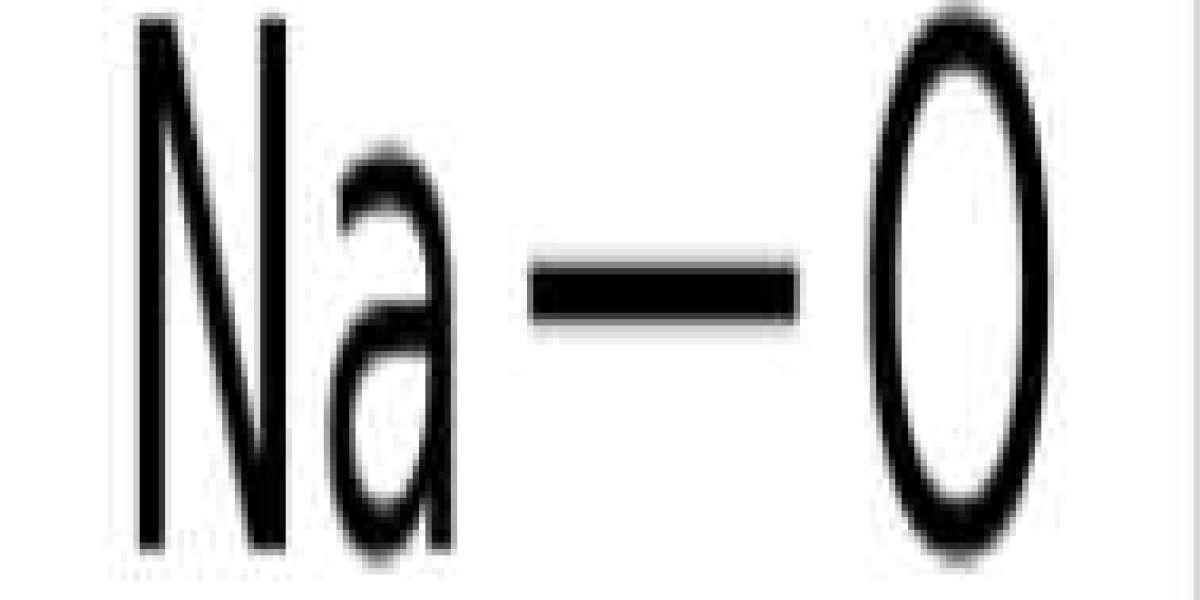Sodium hydroxide, also known as lye and caustic soda,[1][2] is an inorganic compound with the formula NaOH. It is a white solid ionic compound composed of sodium cation Na+ and hydroxide anion OH−.
Sodium hydroxide is a highly caustic base and base that breaks down proteins at ordinary ambient temperatures and can cause severe chemical burns. Very soluble in water, easy to absorb moisture and carbon dioxide in the air. Formation of a series of hydrates NaOH·nH2O[11] The monohydrate NaOH·H2O crystallizes from aqueous solutions between 12.3 and 61.8 °C. Commercially available "sodium hydroxide" is usually this monohydrate and published data may refer to it rather than to the anhydrous compound.
One of the simplest hydroxides, sodium hydroxide is often used with neutral water and acidic hydrochloric acid to demonstrate pH to chemistry students. [12]
Sodium hydroxide is used in many industries: in the manufacture of wood pulp and paper, textiles, drinking water, soaps and detergents, and as a drain cleaner. Global production in 2004 was about 60 million tons, while demand was 51 million tons.
Pure sodium hydroxide is a colorless crystalline solid that melts without decomposition at 318 °C (604 °F) and has a boiling point of 1,388 °C (2,530 °F). It is very soluble in water, and has low solubility in polar solvents such as ethanol and methanol [14]. Sodium hydroxide is insoluble in non-polar solvents such as ether.